Crystallography - Defining the Shape of Our Modern World
Book Exhibit Gallery
In chronological order
THEOPHRASTUS, (ca. 372 B.C. - 287 B.C.). "De Lapidibus" in Eis Organon Aristotelous [Opera Graece]. 5 vols. Venice: Aldus Manutius, 1495-1498.
FIRST PRINTED EDITION of the earliest surviving work mentioning crystals. De Lapidibus (On Stones), written by the Greek scholar Theophrastus, deals primarily with mining and the uses of various ores, but it also gives descriptions of many kinds of crystalline minerals. "It remained for 1800 years one of the most authoritative treatise on minerals, referenced and quoted by writers down to modern times. . . . Theophrastus is in fact the first investigator to treat mineral substances for themselves rather than for their magical or curative properties, and he may be considered the best and most accurate mineralogist among the ancients."
Schuh, Curtis P. Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p1407.
GAIUS PLINIUS SECUNDUS. (23 - 79 A.D.). Naturalis Historiæ. Venice: Rainaldi de Nouimagio, 1483.
Considered even more influential than Theophrastus's De Lapidibus, Naturalis Historiæ (Natural History) "is the first printed book dealing with natural history, being an encyclopedia of the ancient world covering many subjects "written by the Roman philosopher Gaius Plinius Secundus, known in English as Pliny the Elder. Chapters 33-37 contain "a comprehensive catalog of minerals and precious stones, including the uses of these materials in medicine, architecture, and especially art." The first printed edition of Pliny's Naturalis Historiæ, which appeared in 1469, was the first science text ever to be set up in type, only 15 years after Johannes Gutenberg (ca. 1398 - 1468) produced his famous bible.
Schuh, Curtis P. Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p1156-1157.
ALBERTUS MAGNUS (1193 - 1280). De Mineralibus Libri Quinque. Augsburg: Sigmund Grim[m] and Marx Wirsung, 1519.
Albertus Magnus's De Mineralibus (On Minerals) is the most important medieval tract on mining and metallurgy, with significant observations on precious stones and alchemy. It is important for its role in the transformation of mineralogy into a full-fledged science. "The tradition of magical and curative powers in gems still permeates the text, but one feels the author fighting the superstitions of the ancient beliefs, and seeing the world in a new, more skeptical and practical view." Included in the exhibit is a copy of the seventh edition, from 1519. The first printed edition had appeared in 1476.
Schuh, Curtis P. Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p35.
More information on this book from the above cited reference.
GEORG AGRICOLA (1494 - 1555). De Ortu et Causis Subterraneorum [and other works]. Basel: Hieronymus Frobenius and Nikolaus Episcopius, 1546.
IRST EDITION of the first handbook of modern systematic mineralogy. The major portion of this collected edition of five important works by Georg Agricola on mineralogy, geology, and mining is "a refutation of the geological ideas of the ancient philosophers, the alchemists, and the astrologers. It displays Agricola's wide ranging knowledge of the classic authors and his keen observational and logical reasoning ability." Interestingly, Isaac Newton's personal copy of the first edition of this book was sold to a pawn shop in Las Vegas last year (for a ridiculously low price!); the sale was featured on an episode of the television show Pawn Stars.
Schuh, Curtis P. Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p20.
More information on this book from the cited reference above.
JOHANNES KEPLER (1571 - 1630). "Strena seu de Nive Sexangula" in Amphitheatrum Sapientiae Socraticae Joco-seriae, Caspar Dornau, ed. Hanau: Daniel and David Aubrii & Clementis Schleichius, 1619.
"Kepler's Strena Seu de Nive Sexangula is the first scientific treatise concerning crystallography." In 1611, the German astronomer Johannes Kepler (discoverer of the three laws of planetary motion) wrote a small pamphlet as a present to his friends entitled Strena seu de Nive Sexangula (New Year's Gift, or on Hexagonal Snow). Kepler liked to look for hidden regularities and mathematical order in the material world. As an amusement, Kepler proposed the idea that the beautiful hexagonal structure of snowflakes was due to a regular geometric arrangement of tiny solid subunits. He discussed simple cubic and hexagonal packings of spheres, but ultimately was unable to explain the shapes of snowflakes based on geometry alone.
Schuh, Curtis P. Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p832.
More information on this book from the cited reference above.
ROBERT HOOKE (1635 - 1703). Micrographia, or, Some Physiological Descriptions of Minute Bodies made by Magnifying Glasses. With Observations and Inquiries Thereupon. London: Printed by J. Martyn and J. Allestry, 1665.
FIRST EDITION of this landmark work by Robert Hooke, one of the most influential scientific books of the 17th century. The observations were made with Hooke's newly perfected compound microscope, enabling the study of inorganic matter such as crystals, as well as investigations of plant and animal life such as the honeycomb structure of cork and the anatomy of a flea. In fact, in this book Hooke coined the word 'cell' to describe the basic microscopic units of tissue.
Geoffrey Keynes, A Bibliography of Dr. Robert Hooke. Gloucestershire: Clarendon Press, 1960.
More information on this book from the cited reference above.
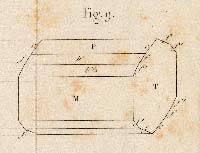
NICOLAUS STENO (1638 - 1686). De Solido intra Solidum. Florence: Ex Typographia sub Signo Stellæ, 1669.
FIRST EDITION of probably the earliest crystallographic observations, made by the Danish physician Niels Stensen (who wrote under his Latinized name Nicolaus Steno). In 1669, Stensen observed in his book De Solido intra Solidum (On Solids within Solids) that the angles between the faces of a crystal of quartz were always the same, even for crystals of different shape or size. Stensen correctly speculated that this constancy was due to some kind of regularity of the internal structure of the crystal.
Schuh, Curtis P. Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p1381-1382.
More information on this book from the cited reference above.
JEAN BAPTISTE ROM DE L'ISLE (1736 - 1790). Cristallographie. Paris: Chez Didot jeune [etc.], 1783.
FIRST EDITION of the book by Jean Baptiste Romé de l'Isle to contain his law of constancy of interfacial angles. This law states that crystals of the same substance show the same angles between corresponding faces, even though they might have different overall shapes such as plates or needles. These angles are therefore characteristic for crystals of each substance. Romé de l'Isle'sCristallographie ranks as one of the great contributions to the science of crystals, in which he attempted to devise a comprehensive classification scheme based on crystal shapes.
Curtis P. Schuh, Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p1256-1257.
More information on this book from the cited reference above.
RENÉ JUST HAÜY (1743 - 1822). Essai d'une Théorie sur la Structure des Crystaux. Paris: Chez Gogué & Née de la Rochelle, 1784.
FIRST EDITION of the important 1784 book Essai d'une Théorie sur la Structure des Crystaux (Essay on a Theory of the Structure of Crystals) by French mineralogist René Just Haüy in which he independently rediscovered Johannes Kepler's idea that crystals are composed of identical building blocks in a regular arrangement. Haüy went much farther, however, in determining the consequences of this idea: he found that each crystal face could be described by a set of three numbers, which were either infinity or small integers such as 1, 2, 3, . . . up to about six. Haüy called this mathematical regularity the law of rational indices.
Diana H. Hook, Jeremy M. Norman, Haskell F. Norman Library of Science and MedicineSan Francisco: Jeremy Norman Co., 1981, item 1021.
Curtis P. Schuh, Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p673-674.
More information on this book from the cited reference above.
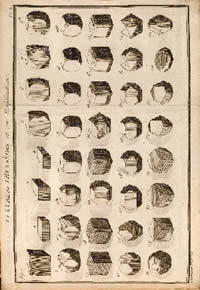
JAMES SOWERBY (1757 - 1822). British Mineralogy: or Coloured Figures intended to elucidate the Mineralogy of Great Britain. London: Printed by R. Taylor (volume 5 by Arding & Merrett) and sold by the Author, J. Sowerby, and by White, (1802-) 1804-1817.
"FIRST EDITION of the supreme work of British topographical mineralogy and the most ambitious color plate work on mineralogy ever published." (1) James Sowerby makes clear in the preface to his work that all the splendid plates were drawn and engraved after actual mineral specimens. "British Mineralogy . . . contains the largest number of plates and some of the finest examples of hand-colored mineral illustration ever produced. . . . Its 550 engraved, hand-colored plates not only depict the mineral wealth of Great Britain, but each plate is accompanied by descriptive text that explains the pictured minerals in a casual style of someone who knew and appreciated minerals." (2)
(1) Conklin, Lawrence H. "James Sowerby, his Publications and Collections," The Mineralogical Record 1995, 26, 85-105.
(2) Schuh, Curtis P. Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p1366.
More information on this book from the cited references above.
MORITZ L. FRANKENHEIM (1801 - 1869).Crystallonomische Aufsätze. (Isis von Oken 1826, columns 497-515 and 542-565.)
FIRST EDITION, JOURNAL ISSUE, of Moritz Frankenheim's discovery of the existence of exactly thirty two kinds of crystal shapes, published in the scientific journal Isis von Oken in 1826. Frankenheim's case is, unfortunately, a classic example of how sometimes an important discovery can be published but largely ignored, and credit given instead to someone later who came to the same conclusion. In fact, Frankenheim's results were subsequently reproduced by two other scientists, both times without mentioning the precedent: first in 1830 by the German mineralogist Johann Hessel (1796 - 1872), and then in 1850 by the French mathematician Auguste Bravais.
Curtis P. Schuh, Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p540-541.
More information on this book from the cited reference above.
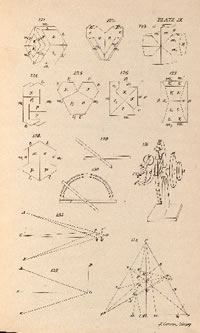
WILLIAM MILLER (1801 - 1880). Treatise on Crystallography. Cambridge: For J. & J.J. Deighton, [etc.], 1839.
FIRST EDITION of the 1839 work Treatise on Crystallography by the Welsh mineralogist William Miller, which popularized a better method to describe the relative orientations of crystal faces in three dimensions. The advantages of "Miller Indices", as they became known, to describe the orientations of crystal faces were almost immediately recognized. None of the previous notational schemes were as easy to apply or as widely accepted as this method, which is still used today."
Schuh, Curtis P. Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p1045.
More information on this book from the cited reference above.
LOUIS PASTEUR (1822 - 1895). Recherches sur les Relations qui peuvent Exister entre la Forme Cristalline, la Composition Chimique et le Sens de la Polarisation Rotatoire. (Annales de Chimie et de Physique, 1848, 24, 442-459.)
FIRST EDITION, JOURNAL ISSUE, of this famous paper which Louis Pasteur presented to the Academy of Sciences on 9 October 1848, entitled Research on the Relations that can Exist between Crystalline Form, Chemical Composition, and the Sense of Rotary Polarization. In this work, Pasteur "provided the impetus and techniques for an entirely new approach to the study of chemical structure and composition. In essence, Pasteur opened the way to a consideration of the disposition of atoms in space, -what we call today the science of stereochemistry."
Complete Dictionary of Scientific Biography. Detroit: Charles Scribner's Sons, 2008, v10, p350-416.
J. R. Partington, A History of Chemistry, New York: MacMillan, vol. 4, 1964, p750-752.
More information on this book from the cited references above.
AUGUSTE BRAVAIS (1811 - 1863). Mémoire sur les Systèmes Formeés par des Points Distribués Regulièerement sur un Plan or dans d'Espace (J. Ecole Polytech. 1850, 19, 1-128.)
FIRST EDITION, JOURNAL ISSUE, of the paper in which the French physicist and mineralogist Auguste Bravais (1811 - 1863) addressed the question "What shape can a crystal building block have and still pack in three dimensions?" In this 1850 journal article Memoir on the Systems formed by Points Regularly Distributed on a Plane or in Space, Bravais proposed 14 types of crystal building blocks now called Bravais lattices.
Curtis P. Schuh, Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p256-257.
More information on this book from the cited reference above.
EVGRAF STEPANOVICH FEDOROV (1853 - 1919). Symmetry of Crystals (American Crystallographic Association Monograph No. 7), David and Katherine Harker (transl.), [Buffalo, N.Y.: American Crystallographic Association, 1971], pp. 50-131.
FIRST EDITION IN ENGLISH of E. S. Fedorov's classic work, which provides the answer to the question, "How many kinds of symmetry can a crystal have?" Fedorov showed that there are exactly 230 kinds of symmetry, now called space groups. Fedorov shares this achievement with the German mathematician Arthur Schoenflies and the British mineralogist William Barlow, each of whom independently came to the same conclusion.
Curtis P. Schuh, Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p555-556.
More information on this book from the cited reference above.
ARTHUR SCHOENFLIES (1853 - 1928). Krystallsysteme und Krystallstructur. Leipzig: B.G. Teubner, 1891.
FIRST EDITION of this important work by Arthur Schoenflies, which presents his independent enumeration of the 230 different kinds of symmetry that crystals can possess. This result was achieved independently and almost simultaneously by Schoenflies, the Russian mineralogist E. S. Fedorov, and the English amateur scientist William Barlow.
P. P. Ewald, Ed., Fifty Years of X-Ray Diffraction Utrecht: International Union of Crystallography, 1962, p352.
More information on this book from the cited reference above.
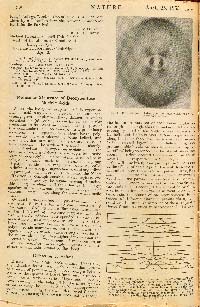
WILLIAM BARLOW (1845 - 1934). Probable Nature of the Internal Symmetry of Crystals. (Nature 1883, 29, 186-188).
FIRST EDITION, JOURNAL ISSUE of the 1883 journal article Probable Nature of the Internal Symmetry of Crystals by the English amateur scientist William Barlow, in which he guessed crystal structures for table salt (sodium chloride) and several related substances, based on ideas about how spheres might pack in space. Although largely ignored for lack of evidence for many years, in 1913 Barlow's suggested crystal structure for table salt was proven by X-ray diffraction to be correct.
"William Barlow, Crystallography 1845 - 1934," J. Chem. Soc. 1935, 1328-1330.
More information on this book from the cited reference above.
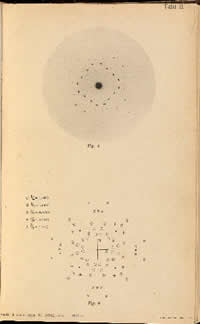
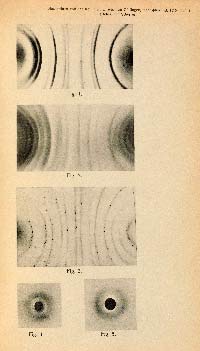
MAX VON LAUE (1879 - 1960). Interferenzerscheinungen bei Röntgenstrahlen. (Sitzungsberichte der Königlich Bayerischen Akademie der Wissenschaften, Mathematische-Physische Klasse 1912, 42, 303-322.)
FIRST EDITION, JOURNAL ISSUE, of Max von Laue's landmark paper Interferenzerscheinungen bei Röntgenstrahlen (Interference Phenomena of Roentgen Rays)announcing the discovery of the diffraction of X-rays by crystals. In a postcard to Laue, dated 10 Jun 1912, Albert Einstein (1879 - 1955) called the discovery "one of the most beautiful experiments physics has ever seen." Laue's discovery earned him the 1914 Nobel Prize in physics.
Complete Dictionary of Scientific Biography. Detroit: Charles Scribner's Sons, 2008, v8, p50-53.
More information on this book from the cited reference above.
WILLIAM L. BRAGG (1890 - 1971). The Diffraction of Short Electromagnetic Waves by Crystals. (Proceedings of the Cambridge Philosophical Society 1913, 17, 43-57.) AND
The Structure of Some Crystals as Indicated by their Diffraction of X-ray. (Proceedings of the Royal Society of London 1913,A89, 248-277.)
FIRST EDITIONS, JOURNAL ISSUES of the announcements by William L. Bragg of two important discoveries: Bragg's law and the first crystal structures determined by X-ray diffraction. In these papers, the father and son team of William H. Bragg and William L. Bragg showed for the first time that the locations of atoms in crystals could be deduced from proper interpretation of the X-ray diffraction pattern; these papers are the founding documents of the science of X-ray crystallography. For these and related researches the father and son team received the Nobel Prize for physics in 1915.
Curtis P. Schuh, Mineralogy & Crystallography: An Annotated Bibliography of Books Published 1469 through 1919. Tucson: privately published, 2005, p247-248.
More information on these articles from the cited reference above.
Page images from The Structure of Some Crystals as Indicated by their Diffraction of X-rays. (Proceedings of the Royal Society of London 1913, A89, 248-277.)
- Page 246 Note by William H. Bragg
- Page 247 Note by William H. Bragg
- Page 254
- Page 255
- Page 264
- Page 265
- Page 284
- Page 285
Page 46 from WILLIAM L. BRAGG (1890 - 1971). The Diffraction of Short Electromagnetic Waves by Crystals. (Proceedings of the Cambridge Philosophical Society 1913, 17, 43-57.)
CHARLES G. DARWIN (1887 - 1962). The Theory of X-ray Reflexion. (Philosophical Magazine 1914, 27, 315-333.)
FIRST EDITION, JOURNAL ISSUE, of Charles G. Darwin's paper The Theory of X-ray Reflexion, which gave mathematical formulas for calculating the intensities of diffracted X-rays, and "which laid the foundation for all subsequent interpretation of X-ray diffraction by crystals. These researches were probably Darwin's most important contribution to theoretical physics." Darwin was the grandson of the evolutionary biologist Charles Darwin (1809 - 1883).
Complete Dictionary of Scientific Biography. Detroit: Charles Scribner's Sons, 2008, v3, p563-565.
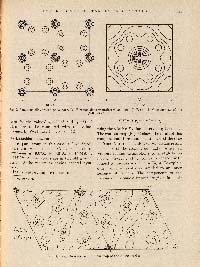
PETER DEBYE (1884 - 1966) and PAUL SCHERRER (1890 - 1969). Interferenzen an Regellos Orientierten Teilchen im Röntgenlicht. (Nachrichten von der Königl. Gesellschaft der Wissenschaften zu Göttingen. Mathematisch-Physikalische Klasse 1916, 29, 1-15.)
FIRST EDITION, JOURNAL ISSUE of the discovery of Peter Debye and Paul Scherrer that X-ray diffraction could be used to determine the crystal structures of powdered samples. "Their motivation was to avoid the problem of growing the large single crystals that were needed to use the methods developed by Max von Laue and the Braggs, and to facilitate the cumbersome data evaluation processes associated with these methods." Their method, which was published in their paper Interference of Randomly Oriented Particles in X-ray Light in 1916, "was independently developed around the same time by Albert Hull (1880 - 1966) at the General Electric Research Laboratory."
Holzwarth, Uwe and Gibson, Neil. "The Scherrer Equation versus the 'Debye-Scherrer Equation'," Nature Nanotechnology 2011, 6, 534.
More information on the authors from the cited reference above
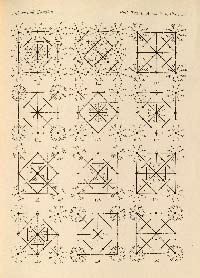
RALPH W. G. WYCKOFF (1897 - 1994). The Analytical Expression of the Results of the Theory of Space Groups. Washington: Carnegie Institution, 1922.
AND
WILLIAM T. ASTBURY (1898 - 1961) and KATHLEEN YARDLEY [LONSDALE] (1903 - 1971). Tabulated Data for the Examination of the 230 Space-Groups by Homogeneous X-Rays. (Philosophical Transactions of the Royal Society of London, 1924, A224, 221-257).
FIRST EDITIONS of Ralph Wyckoff's book The Analytical Expression of the Results of the Theory of Space Groups, and William Astbury and Kathleen Yardley's article Tabulated Data for the Examination of the 230 Space-Groups by Homogeneous X-Rays, which tabulated in a convenient way the positional coordinates of symmetry-related points for each of the 230 space groups. The tables in Wyckoff's book and the figures in Astbury and Yardley's article formed the basis for what eventually became the International Tables for X-ray Crystallography, the first edition of which appeared in 1935.
Jan Drenth and Aafje Looyenga-Vos, "Ralph W. G. Wyckoff," Acta Crystallographica 1995, A51, 649-650.
Complete Dictionary of Scientific Biography. Detroit: Charles Scribner's Sons, 2008, v1, p319-320.
Complete Dictionary of Scientific Biography. Detroit: Charles Scribner's Sons, 2008, v8, p484-486.
More information about the authors from the cited references above
GEORGE L. CLARK (1892 - 1969). Applied X-rays. New York: McGraw-Hill, 1927.
FIRST EDITION of an important book describing the scientific, industrial, and medical uses of X-rays, including many techniques developed by the author, George Clark. In the course of his nearly thirty years of research, Clark used X-ray methods to characterize a wide variety of materials, including metals and minerals, natural and synthetic fibers, natural and synthetic rubber, clays, carbon black, storage battery plates, corks, and waxes. Clark was a long-time professor of chemistry at the University of Illinois at Urbana-Champaign.
"George Lindenberg Clark and High-Intensity X-ray Tubes," American Chemical Society, National Historic Chemical Landmarks website (http://www.acs.org/content/acs/en/education/whatischemistry/landmarks/noyeslaboratory.html#george-lindenberg-clark-biography, accessed 30 Jan 2012).
More information about the author from the cited reference above.
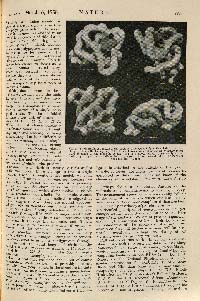
KATHLEEN Y. LONSDALE (1903 - 1971). The Structure of the Benzene Ring. (Nature 1928, 122, 810.)
FIRST EDITION, JOURNAL ISSUE, of the crystallographic proof of the hexagonal structure of the benzene ring, published by the British crystallographer Kathleen Lonsdale in 1928. Although she confessed that she "knew no organic chemistry and very little of any other kind," this finding was one of the most outstanding advances in that subject for many decades. Today, crystallographic investigations of organic molecules such as pharmaceuticals and proteins are crucial to the development of improved medicines and many other scientific advances.
Complete Dictionary of Scientific Biography. Detroit: Charles Scribner's Sons, 2008, v8, p484-486.
More information about the author from the cited reference above.
WILSON A. BENTLEY (1865 - 1931). Snow Crystals. New York: McGraw-Hill, 1931.
FIRST EDITION of one of the most remarkable books ever written by an amateur scientist. From the earliest memories of our childhood, many of us can remember hearing the phrase "no two snowflakes are alike." The scientific evidence in support of this assertion stems largely from the efforts of one man, Wilson Bentley, who took the pictures on his family farm in Jericho, Vermont. Bentley's hope of someday giving to the world a book of his snowflake pictures came to fruition but, tragically, only a few days after the book was published, he died of an attack of pneumonia.
"Wilson Alwyn Bentley," Dictionary of American Biography. New York: Charles Scribner's Sons, 1944.
More information about the author from the cited reference above
ARTHUR LINDO PATTERSON (1902 - 1966). A Fourier Series Method for the Determination of the Components of Interatomic Distances in Crystals. (Phys. Rev. 1934, 46, 372-376.)
FIRST EDITION, JOURNAL ISSUE, of the paper by Lindo Patterson that announced the Patterson function, which remains today an essential tool to determine the locations of the atoms in a crystal from the crystal's X-ray diffraction pattern. Before Patterson's work, crystal structures could be deduced from the diffraction pattern only in very special circumstances involving the simplest structures. "Most of the presently available wealth of information about the structures of solids has been obtained through the application of the Patterson method to crystal-structure analyses. This includes our knowledge of complex biological molecules, such as penicillin, DNA, and hemoglobin."
Anon. "Arthur Lindo Patterson; Crystal Structure Analyst," Physics Today 1967 (Feb.), 97-99.
R. E. Marsh and D. P. Shoemaker, "Arthur Lindo Patterson 1902 -1966," Acta Crystallographica1967, 22, 749-750.
More information about the author from the cited references above.
JEROME KARLE (1918 - 2013) and HERBERT HAUPTMAN (1917 - 2011). The Phases and Magnitudes of the Structure Factors. (Acta Crystallographica 1950, 3, 181-187.)
FIRST EDITION, JOURNAL ISSUE, of the announcement of a powerful new method to determine the structures of crystals. The "direct method," developed by Herbert Hauptman and Jerome Karle beginning in 1950, is in many respects complementary to the Patterson method but applicable to a wider variety of crystals. "By the mid 1980s the atomic structures of approximately 40,000 substances had been determined through use of direct methods, as compared to only some 4,000 determined by other methods in all the years prior to about 1970. Hauptman and Karle were awarded the 1985 Nobel Prize in chemistry for this discovery."
World of Chemistry, Gale, 2006, "Herbert A. Hauptman," Gale Biography in Context, Web. 26 Jan 2012.
"Jerome Karle," World of Scientific Discovery. Detroit: Gale, 2006.
"Herbert Hauptman," Encyclopedia of World Biography. Vol. 24. Detroit: Gale, 2005.
More information about the authors from the cited references above.
JAMES D. WATSON (1928 -) and FRANCIS H. C. CRICK (1916 - 2004). Molecular Structure of Nucleic Acids. A Structure for Deoxyribose Nucleic Acid. (Nature, 1953, 171, 737-738.) WITH:
MAURICE H. F. WILKINS (1916 - 2004), ALEXANDER R. STOKES (1919 - 2003), and HERBERT R. WILSON (1929 - 2008). Molecular Structure of Deoxypentose Nucleic Acids. (Nature, 1953, 171, 738-740.) AND WITH:
ROSALIND E. FRANKLIN (1920 - 1958) and RAYMOND G. GOSLING (1926 -). Molecular Configuration in Sodium Thymonucleate. (Nature, 1953, 171, 740-741.)
FIRST EDITION, JOURNAL ISSUE, of the most important twentieth-century discovery in biology, and one of the most important in the whole of science, the identification of the double helix structure of DNA. This discovery, which had far-reaching implications was recognized by the award of the 1962 Nobel prize in medicine to James Watson, Francis Crick, and Maurice Wilkins. Rosalind Franklin, who many believe deserved a share of the prize, had died four years earlier; the Nobel prize is not awarded posthumously.
DOROTHY HODGKIN (1910 - 1994) [et al]. X-ray Crystallographic Evidence on the Structure of Vitamin B12. (Nature 1954, 174, 1169-1171.)
FIRST EDITION, JOURNAL ISSUE of Dorothy Hodgkin's 1954 initial determination of the structure of vitamin B12, a dietary component important to human health. For her contributions to the X-ray analysis of natural products such as steroids, antibiotics, and proteins, Hodgkin was awarded the Nobel Prize in Chemistry for 1964. William L. Bragg described the B12 study as the crystallographic equivalent to "breaking the sound barrier".
Complete Dictionary of Scientific Biography. Detroit: Charles Scribner's Sons, 2008, v21, p333-339.
Guy Dodson, "Dorothy Mary Crowfoot Hodgkin O.M. 12 May 1910 - 29 July 1994", "Biographical Memoirs of Fellows of the Royal Society" 2002, 48, 179-219.
More information about the author from the cited references above.
JOHN KENDREW (1917 - 1997). A Three-Dimensional Model of the Myoglobin Molecule Obtained by X-Ray Analysis. (Nature 1958, 181, 662-666.)
FIRST EDITION, JOURNAL ISSUE, of the paper announcing the first solution of the three-dimensional molecular structure of a protein, myoglobin. Myoglobin is a component of the hemoglobin that carries oxygen in the bloodstream. For this achievement, John Kendrew received the 1962 Nobel Prize in chemistry together with his friend and colleague Max Perutz (1914 - 2002), who solved the structure of the related and more complex protein, hemoglobin, two years after Kendrew's achievement.
Jeremy Norman, "The First Solution of the Three-Dimensional Molecular Structure of a Protein (1958 - 1960)," http://www.historyofinformation.com/expanded.php?id=3015, accessed 5 Jan 2012.